
The Spark and the Fire: Why Atrial Fibrillation Is Not the Disease
Atrial Fibrillation: It’s Not Just a Heart Rhythm Problem
Dr John Sciales, Director: CardioCore Metabolic Wellness Center
Summary
Atrial fibrillation is not just a problem of heart rhythm. It is a signal that something deeper is happening within the body.
Most care focuses on controlling the rhythm, but the rhythm is not the disease. Beneath the surface lies a metabolic and inflammatory process that affects the heart, blood vessels, and multiple organ systems over time.
Until that underlying terrain is understood and addressed, the disease continues to progress. This is not about chasing symptoms. It is about identifying the cause.
The Warning Sign Most People Miss
Atrial fibrillation, often called AFib, is one of the most common heart rhythm problems in the world today. Millions of people are living with it, and the number continues to rise year after year. In fact, AFib has become so common that it is now considered a growing public health issue, closely tracking the rise in obesity, diabetes, and high blood pressure (1,2).
Most people hear the same explanation when they are diagnosed. They are told that their heart rhythm is irregular, that the rate needs to be controlled, and that a blood thinner may be necessary to reduce the risk of stroke. All of this is important, and in many cases, life-saving.
But it is not the full story.
Because atrial fibrillation is not simply a problem of rhythm. It is a signal, a warning, that something deeper has been developing in the body, often for years before the first symptom appears.
What makes AFib so confusing is that it feels sudden. A person may experience palpitations, fatigue, or shortness of breath, and it seems as though the condition came out of nowhere. But biologically, that is almost never the case.
AFib does not begin the day you feel it.
It begins much earlier, quietly, as gradual changes take place within the body, and more importantly, within the cells of the heart itself.
The Heart’s Electrical System Is Made of Living Cells
We often describe AFib as an electrical disorder, and that is true to a point. The rhythm becomes chaotic because the electrical signals in the atria are disorganized. But what is often overlooked is that this electrical system is not made of wires.
It is made of living, functioning cells.
These cells depend on a stable internal environment. They rely on proper blood flow, balanced signaling, and metabolic health to maintain normal electrical activity. They are not separate from the rest of the body. They are influenced by the same biological forces that affect the arteries, the immune system, and metabolism as a whole.
Over time, certain conditions begin to change these cells. Insulin resistance develops quietly, often years before blood sugar or hemoglobin A1c becomes abnormal. As this happens, a chronic, low-grade inflammatory state begins to circulate throughout the body. Weight gain, especially around the abdomen called visceral fat, reflects this deeper metabolic imbalance. Blood pressure rises as vascular function declines. Sleep becomes disrupted, and stress signaling remains chronically activated.
In many cases, this proinflammatory state is first triggered by insulin resistance, long before traditional markers of disease are detected. By the time glucose or A1c levels become elevated, the underlying process has often been present for years, already affecting the heart, blood vessels, and multiple organ systems.
By the time the numbers change, the biology has already been at work for years.
These are often labeled as “risk factors.” But that label minimizes what they actually are. They are active biological processes that slowly reshape the structure and function of the heart.
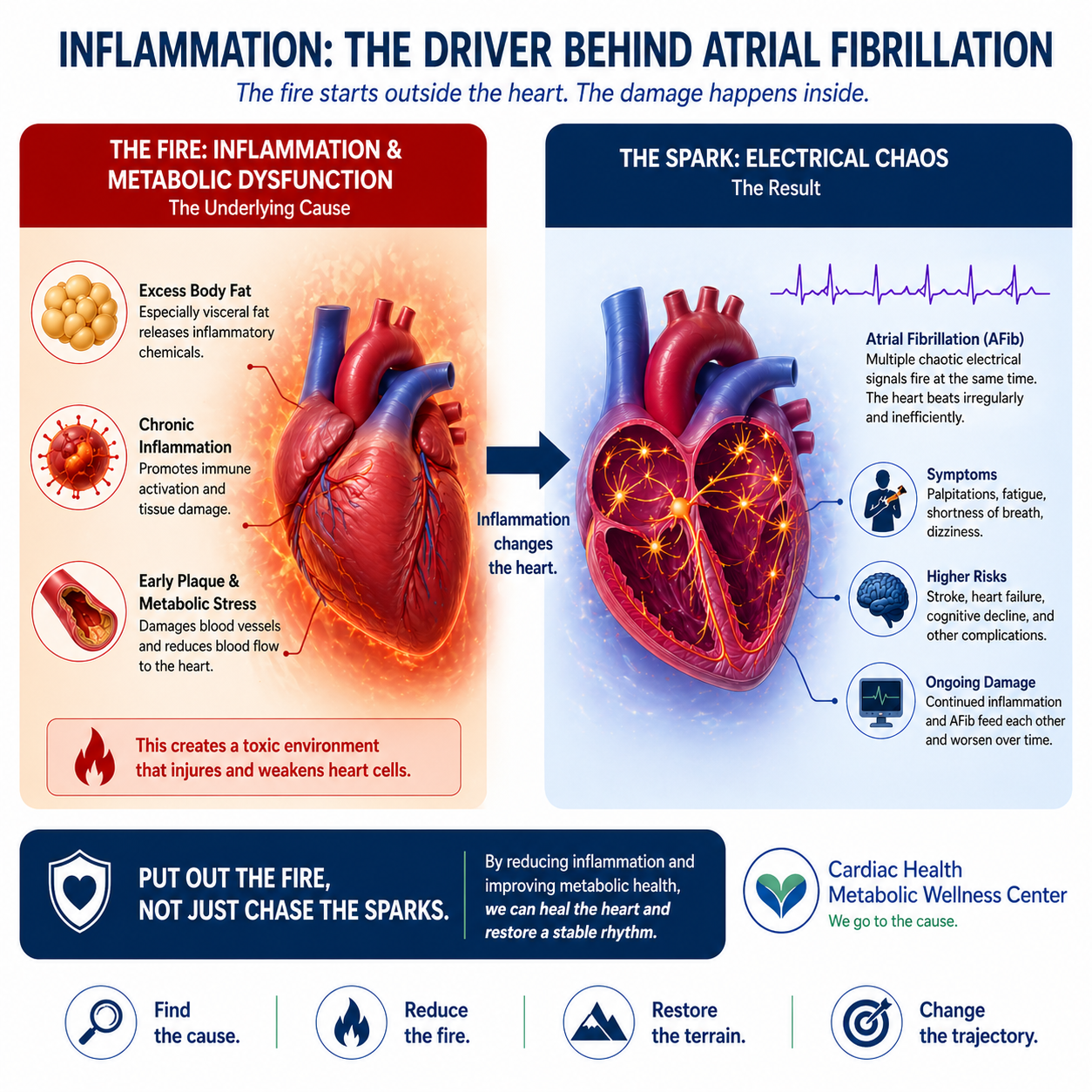
Figure 1.
The heart’s electrical system is made up of living cells. In normal sinus rhythm, these cells generate and conduct a single, organized electrical impulse that produces a coordinated heartbeat. In atrial fibrillation, these same cells become electrically unstable, generating multiple disorganized signals that lead to a chaotic and irregular rhythm.
What Happens Beneath the Surface
As these processes continue, the heart begins to remodel itself. This is not something a person can feel at first. It happens gradually, beneath the surface.
Inflammatory signals begin to affect the heart muscle, promoting subtle injury and repair. Over time, this leads to the development of fibrosis, small areas of scar tissue that disrupt the normal architecture of the atria. At the same time, fat begins to accumulate around the heart, particularly in the epicardial space, where it acts as a source of inflammatory signaling directly adjacent to the heart muscle (3,4).
The cells that once conducted electrical signals in a smooth, coordinated way now operate in an environment that is inflamed, structurally altered, and metabolically unstable. Electrical conduction becomes inconsistent. Signals fragment. Pathways become disrupted.
This creates the ideal conditions for electrical disorganization. And eventually, atrial fibrillation appears. Not as the beginning of disease, but as the moment the disease finally becomes visible.
Why This Matters More Than You Think
This leads to a critical insight that changes how we should think about AFib.
Most people do not develop atrial fibrillation in an otherwise healthy heart. They develop it in a heart that has already been under metabolic stress for years.
That stress is driven by the same biological processes that underlie many of the most common chronic diseases today. The same processes that contribute to plaque formation in the arteries, to stiffening of the heart muscle, and to progressive cardiovascular disease are also affecting the cells responsible for maintaining normal rhythm (2,5).
Which means atrial fibrillation is rarely an isolated problem. It is usually part of a much larger picture.
A Question That Changes Everything
When someone is diagnosed with AFib, the focus is almost always on controlling the rhythm.
But a more important question often goes unasked:
Why did the heart become unstable in the first place?
Because if that question is not addressed, we are only treating what we can see, and not what is driving the disease.
Why Treating the Rhythm Isn’t Treating the Disease
Once atrial fibrillation is diagnosed, the focus of care becomes very clear: control the rhythm, control the rate, and reduce the risk of stroke These treatments are important. They can improve how a person feels, reduce immediate risks, and in many cases stabilize the condition. But there is a deeper issue in how atrial fibrillation is approached.
We are becoming increasingly skilled at managing the rhythm of the heart…
without fully addressing the disease that caused the rhythm to break down in the first place. This leads to an important distinction:
The rhythm is not the disease.
And in many cases, treating the rhythm is not treating the disease.
The Rise of Rhythm-Based Treatments
In recent years, there has been a strong shift toward restoring normal rhythm as early as possible.
Cardioversion is now commonly used to reset the heart back into a normal rhythm. Catheter ablation has also become more advanced and more widely used, targeting areas of abnormal electrical activity in the atria.
For many patients, these treatments can be very effective in the short term. The rhythm normalizes, symptoms improve, and there is a sense that the problem has been corrected. But what happens after that is where the limitation becomes clear.
Why Atrial Fibrillation So Often Returns
Atrial fibrillation frequently comes back. Not because the procedures are done incorrectly, but because the underlying environment of the heart has not changed.
If the heart tissue remains inflamed, if fibrosis is already present, if metabolic dysfunction continues to affect the cells, then the same conditions that created atrial fibrillation are still in place (6,7).
In that setting, restoring rhythm is temporary. The system has been reset, but not repaired.
We Reset the Rhythm, But Not the Biology
This is the central issue.
Cardioversion changes the rhythm. Ablation modifies electrical pathways. But neither of these treatments directly reverses the metabolic and inflammatory processes that altered the heart in the first place.
The disease is still there.
The cells are still exposed to the same biological signals, insulin resistance, inflammation, and structural stress, that led to the problem. And over time, the same pattern often returns.
This is why recurrence after cardioversion and even after ablation remains common, particularly in patients with ongoing metabolic dysfunction (8).
Understanding the Role of the Electrophysiologist
Many patients with atrial fibrillation are referred to a cardiologist who specializes in heart rhythm disorders, an electrophysiologist.
Electrophysiologists are highly trained and play an essential role in managing arrhythmias. Their expertise is in diagnosing and treating the electrical system of the heart, often using advanced procedures like ablation.
But their focus is, by design, on the rhythm.
Most patients are not aware that cardiology includes different areas of specialization. As a result, they may assume that all aspects of their heart health are being addressed when they see a heart specialist.
In reality, the evaluation of broader cardiovascular disease, such as atherosclerosis, metabolic dysfunction, and vascular inflammation, may not be the primary focus during rhythm-based care.
This is not a flaw in the system, it is a reflection of how modern medicine is structured. As care has become more specialized, physicians have developed deep expertise in treating well-defined problems like atrial fibrillation.
But in that process, the broader view of the patient is often lost. The rhythm is treated with precision, while the disease that created it continues to progress.
But it does create a gap. Because atrial fibrillation is often not just an electrical problem. It is part of a larger cardiometabolic disease process that extends beyond the rhythm itself.
The Risk That Continues Beneath the Surface
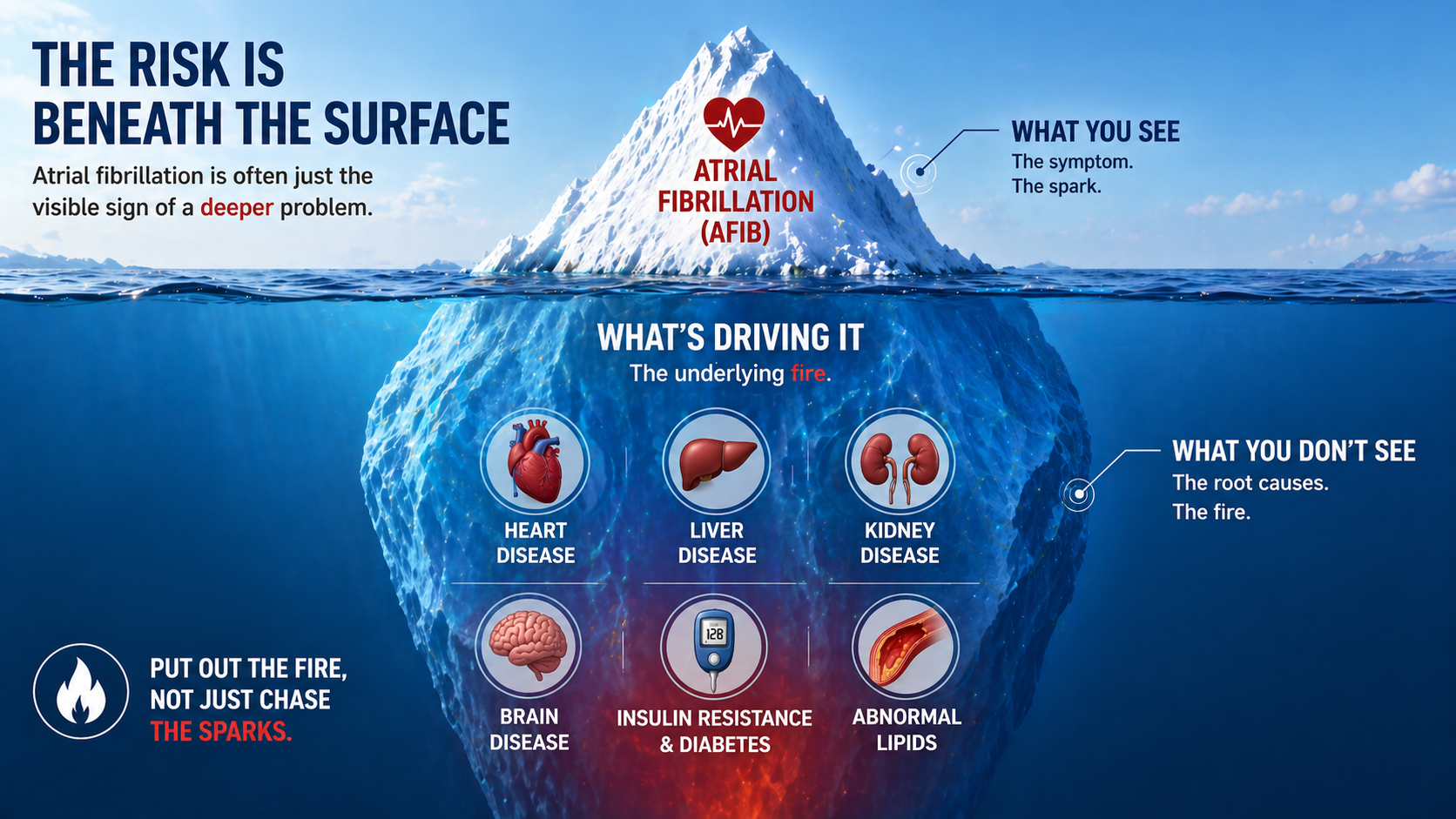
Figure 2.
Atrial fibrillation is the visible tip of the iceberg. Beneath the surface lies the metabolic and inflammatory terrain—insulin resistance, chronic inflammation, and multi-organ disease—that drives both the rhythm disturbance and long-term cardiovascular risk.
Treating the tip without addressing the base leaves the disease untouched.
There is another important truth that patients are rarely told clearly. Most people with atrial fibrillation do not die from the arrhythmia itself.
With proper management, stroke risk can be reduced and heart rate can be controlled. The immediate risks related to the rhythm can be managed effectively. But the underlying disease continues.
The same biological processes that contribute to atrial fibrillation, insulin resistance, inflammation, and vascular dysfunction, are also driving coronary artery disease, heart failure, and long-term cardiovascular risk (9).
So while attention is focused on the rhythm, the broader disease often continues to progress silently.
A Necessary Shift in Thinking
This leads to a critical shift in perspective. Instead of asking only how to control the rhythm, we need to ask:
What is the disease that caused the rhythm to become unstable?
Because if that disease is not addressed, long-term outcomes are unlikely to improve in a meaningful way. Atrial fibrillation is not just something to control. It is something to understand.
Looking Deeper, Putting Out the Fire, Not Just Chasing the Sparks
By the time atrial fibrillation appears, the body has already been under stress for years.
The rhythm is not the beginning of the problem.
It is one of the signals.
And if we want to change long-term outcomes, we have to look beyond the rhythm and begin to understand what is driving it.
Why Traditional Testing Often Misses the Problem
Many people with atrial fibrillation are told that their tests are “normal.”
Their cholesterol looks acceptable.
Their blood sugar and hemoglobin A1c are within range.
Their thyroid and kidney function appear fine.
But this can give a false sense of security.
Most routine lab tests are designed to detect disease once it is already established, not when it is developing. Even commonly used cardiac stress tests are designed to identify advanced blockages that limit blood flow. They are not sensitive for early atherosclerosis or the underlying disease in the artery wall (10,11).
This is why someone can have a normal stress test and still have significant cardiovascular risk.
If we only look for late-stage disease, we miss the opportunity to intervene early, when it matters most.
Why Direct Imaging Changes the Conversation
To truly understand cardiovascular risk, we often need to look directly at the arteries.
Coronary calcium scoring and coronary CT angiography allow us to see plaque within the artery wall long before it causes obstruction. These tools provide insight into disease that cannot be detected by stress testing alone (12).
Because the lumen, the opening of the artery, is not the disease. The disease is in the wall. It is driven by inflammation, metabolic dysfunction, and vascular injury that develop silently over time.
The same biology that creates plaque in the arteries is also affecting the heart muscle and the electrical cells that lead to atrial fibrillation.
The Real Problem: The Metabolic and Inflammatory Burden
At the center of this is what we call the metabolic terrain. This includes insulin resistance, chronic inflammation, oxidative stress, and abnormal signaling that affect every organ system. When this environment is unhealthy, it does not show up in just one place.
Atrial fibrillation is one signal.
Coronary artery disease is another.
Heart failure, kidney disease, liver dysfunction, and even cognitive decline can all emerge from the same underlying process.
These are not separate diseases. They are different expressions of the same biological fire. Research consistently shows that insulin resistance and inflammation are key drivers of both atherosclerosis and atrial remodeling, linking metabolic dysfunction directly to cardiovascular disease and arrhythmias (13,14).
Looking Beneath “Normal” Labs
To find the root cause, we have to go beyond routine testing. We need to understand how the body is functioning long before standard markers become abnormal.
That means evaluating insulin response through an oral glucose tolerance test, not just fasting glucose or A1c. It means recognizing patterns like an elevated triglyceride-to-HDL ratio, which reflects early metabolic dysfunction. It means measuring ApoB to assess the true burden of atherogenic particles, and high-sensitivity CRP to evaluate underlying inflammation.
These markers reveal what is happening beneath the surface.
They show us the fire, not just the sparks.
The Role of Rhythm Specialists, and the Missing Piece
Electrophysiologists play a critical and valuable role in managing atrial fibrillation. They are experts in the electrical system of the heart and in procedures like cardioversion and ablation.
But their focus is, by necessity, on the rhythm.
They reset the system.
They stabilize the rhythm.
They manage the electrical problem.
But the underlying metabolic and inflammatory drivers often remain unaddressed. And this is where the gap exists. Because atrial fibrillation is not just an electrical disorder.
It is a manifestation of a broader cardiometabolic disease process.
Putting Out the Fire
Atrial fibrillation is not the fire. It is one of the sparks.
So is coronary artery disease.
So is heart failure.
So is kidney and liver disease.
These are not separate problems.
They are different expressions of the same underlying process.
If we focus only on the sparks, we never extinguish the source.
We treat what we see, while the real disease continues to burn beneath the surface.
To truly change outcomes, we have to put out the fire. Because if we only treat the spark, the fire keeps burning.
This is what that looks like beneath the surface.
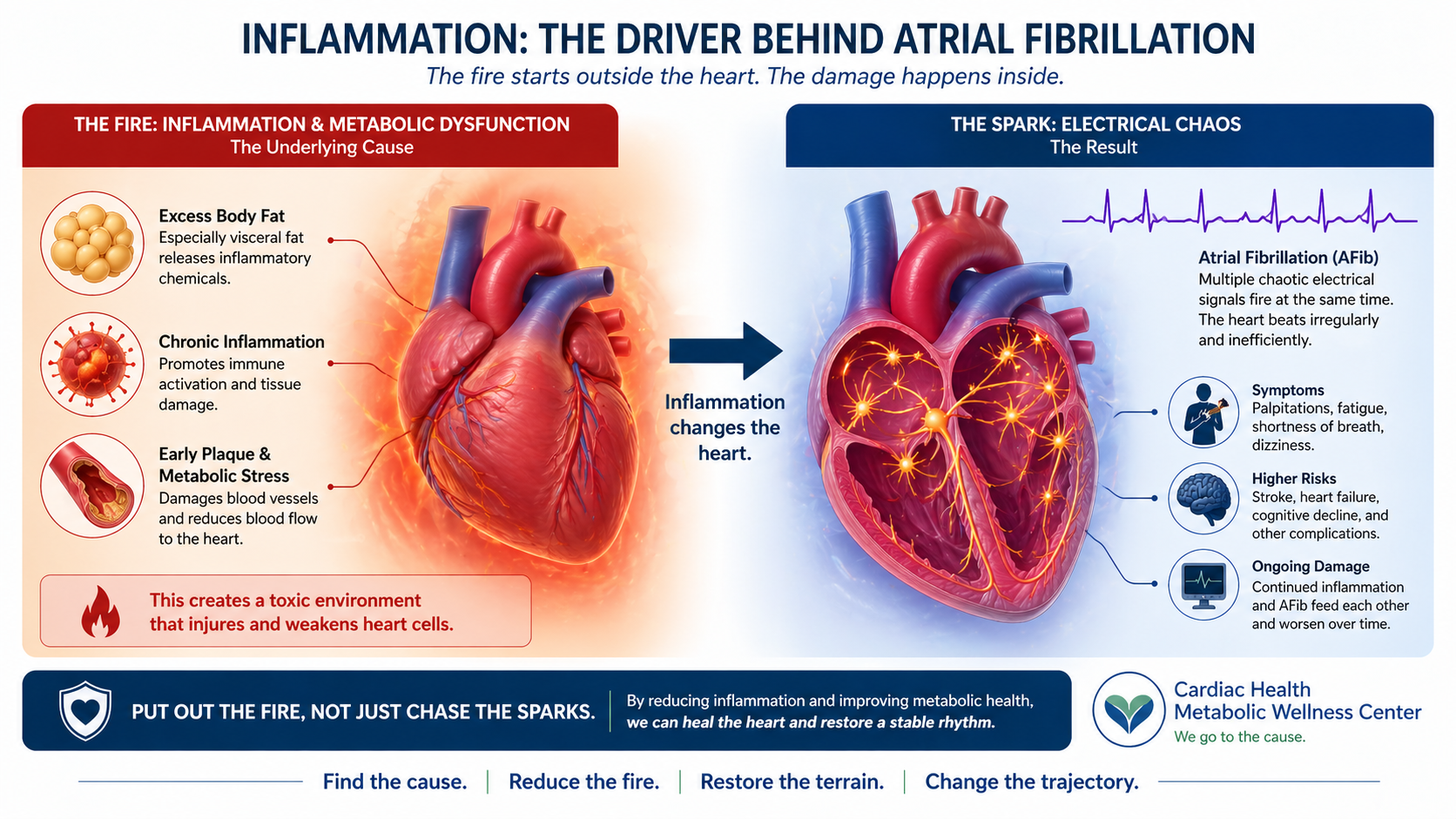
Figure 3.
Chronic inflammation and metabolic dysfunction create a harmful environment that damages the heart’s cells, leading to structural and electrical changes. This underlying “fire” drives instability in the atria, resulting in the disorganized electrical activity seen in atrial fibrillation.
Putting out the fire means identifying and correcting the metabolic terrain that is driving inflammation, vascular disease, and structural changes in the heart. It means looking beyond the rhythm, beyond the symptoms, and understanding the biology that created them.
That is where real change begins.
Looking Deeper: Putting Out the Fire, Not Just Chasing the Sparks
If you have atrial fibrillation, this is your opportunity to look deeper.
Do not stop at controlling the rhythm.
Do not assume normal labs mean everything is fine.
Do not rely on a normal stress test to rule out disease.
Ask what is driving it.
Ask whether there is underlying plaque in your arteries.
Ask whether insulin resistance is present, even if your blood sugar looks normal.
Ask what is fueling inflammation in your body.
Because unless the metabolic terrain is addressed, long-term outcomes are unlikely to improve in a meaningful way.
CardioCore Metabolic Wellness Center
At CardioCore Metabolic Wellness Center, we take a different approach.
We don’t just treat the rhythm.
We don’t just look for blockages.
We identify and treat the biology that drives both.
We look deeper, at your metabolic function, your inflammatory burden, and the early signals of disease that are often missed by routine testing. We focus on identifying problems before they become advanced, and on correcting the underlying drivers of cardiovascular disease at their source.
Because when you change the terrain…
You don’t just manage atrial fibrillation.
You change the direction of your health.
If you have atrial fibrillation, or if you’ve been told your tests are “normal” but something still doesn’t feel right, this is the moment to look deeper.
The most important question is not just how to control your rhythm.
It is:
What is the fire beneath it?
If you’re ready to understand what’s driving your condition and explore a more complete, root-cause approach, I invite you to schedule a consultation.
This is where we begin to move beyond managing disease…
and start changing its course.
Dr John Sciales
Stop guessing. Start seeing what's beneath the Rhythm
Book a Free Discovery Call and Find Out More...
CLICK HERE TO BOOK A DISCOVERY CALL
CLICK HERE TO JOIN OUR PRIVATE CardioCore COMMUNITY
CLICK HERE TO SPEAK TO OUR VIRTUAL ASSISTANT
References
Chugh SS, Havmoeller R, Narayanan K, et al. Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation. 2014;129(8):837–847.
Wang TJ, Parise H, Levy D, et al. Obesity and the risk of new-onset atrial fibrillation. JAMA. 2004;292(20):2471–2477.
Mahajan R, Lau DH, Sanders P. Impact of epicardial adipose tissue on atrial fibrillation. J Am Coll Cardiol. 2015;66(6):667–676.
Al Chekakie MO, Welles CC, Metoyer R, et al. Pericardial fat is independently associated with human atrial fibrillation. J Am Coll Cardiol. 2010;56(10):784–788.
Kannel WB, Wolf PA, Benjamin EJ, Levy D. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: population-based estimates. Am Heart J. 1998;135(5 Pt 1):895–902.
Kotecha D, Lam CSP, Van Veldhuisen DJ, et al. Heart failure with preserved ejection fraction and atrial fibrillation: mechanistic and therapeutic insights. Eur Heart J. 2017;38(20):1559–1567.
Dzeshka MS, Lip GYH, Snezhitskiy V, Shantsila E. Cardiac fibrosis in patients with atrial fibrillation: mechanisms and clinical implications. J Am Coll Cardiol. 2015;66(8):943–959.
Pathak RK, Middeldorp ME, Meredith M, et al. Long-term effect of goal-directed weight management on atrial fibrillation cohort: the ARREST-AF study. J Am Coll Cardiol. 2014;64(21):2222–2231.
Benjamin EJ, Wolf PA, D’Agostino RB, et al. Impact of atrial fibrillation on the risk of death: the Framingham Heart Study. Circulation. 1998;98(10):946–952.
Gibbons RJ, Balady GJ, Bricker JT, et al. ACC/AHA 2002 guideline update for exercise testing. Circulation. 2002;106(14):1883–1892.
Shaw LJ, Berman DS, Maron DJ, et al. Optimal medical therapy with or without percutaneous coronary intervention to reduce ischemic burden: COURAGE trial nuclear substudy. J Am Coll Cardiol. 2008;52(12):913–921.
Budoff MJ, Shaw LJ, Liu ST, et al. Long-term prognosis associated with coronary calcification: observations from a large registry. J Am Coll Cardiol. 2007;49(18):1860–1870.
Grundy SM. Metabolic syndrome update. Circulation. 2008;117(4):e25–e27.
Dzeshka MS, Shahid F, Shantsila A, Lip GYH. Hypertension and atrial fibrillation: an intimate association of epidemiology, pathophysiology, and outcomes. J Am Coll Cardiol. 2017;69(24):2919–2930.
