
The Samurai Sword of Vascular Health How Nattokinase Cuts Through Fibrin, Inflammation, and Cardiometabolic Risk
The Samurai Sword of Vascular Health
How Nattokinase Cuts Through Fibrin, Inflammation, and Cardiometabolic Risk
Dr John Sciales
Director, CardioCore Metabolic Wellness Center
“Getting to the Core… the Path to Wellness- Where Being Healthy Is Not an Accident”
Abstract
Cardiovascular disease is often thought of as a sudden event, such as a heart attack or stroke. In reality, the disease usually develops slowly over many years inside the walls of the arteries. Long before symptoms appear, metabolic dysfunction, inflammation, and endothelial injury begin to alter the biology of the vascular system. Over time these processes lead to plaque formation, plaque instability, and eventually clot formation.
Nattokinase, a proteolytic enzyme derived from the fermented soybean food natto, has attracted attention because of its ability to break down fibrin, a key structural protein in blood clots. Laboratory studies and small human trials suggest that nattokinase can increase fibrinolytic activity and influence markers of clot formation. However, fibrin formation represents one of the final stages of the atherosclerotic process rather than the origin of the disease.
Atherosclerosis progresses through several biological stages beginning with endothelial injury and immune activation, followed by foam cell formation, plaque development, plaque instability, platelet activation, and finally thrombosis. Different therapies act at different points along this timeline. Metabolic therapies, anti-inflammatory agents, lipid-lowering medications, antiplatelet drugs, and fibrinolytic compounds each target distinct steps in the disease process.
This article reviews the biology of atherosclerosis, explains how nattokinase works as a fibrin-degrading enzyme, and places its effects within the broader context of stage-based cardiovascular medicine. While nattokinase may influence the final thrombotic stage of the disease, long-term cardiovascular prevention requires addressing the upstream drivers of atherosclerosis, including metabolic dysfunction, inflammation, and endothelial injury.
Understanding these stages allows clinicians and patients to move beyond treating the final event and instead focus on preventing the biological conditions that make that event possible.
Introduction
Most people believe cardiovascular disease begins with a heart attack. In reality, the disease begins many years earlier within the biology of the arterial wall. Long before symptoms appear, subtle metabolic and inflammatory disturbances begin to reshape the vascular environment. The arteries do not suddenly become diseased overnight. A person does not wake up one morning with a blocked artery or a calcium score of 200. These findings represent the late manifestation of biological processes that have been developing silently for years or even decades. The earliest stage of cardiovascular disease often begins with injury to the endothelium, the delicate inner lining of the blood vessels. This injury may be mechanical, inflammatory, metabolic, or oxidative in origin. Conditions such as insulin resistance, metabolic syndrome, chronic inflammation, and oxidative stress can all disturb endothelial function.[1]
Once the endothelial surface is disrupted, the artery becomes vulnerable to immune cell recruitment and lipid retention within the vessel wall. Circulating monocytes migrate into the artery and differentiate into macrophages. These macrophages ingest retained lipoproteins and transform into foam cells, forming the earliest visible lesion of atherosclerosis.[2] Over time, lipids accumulate, inflammatory signals intensify, and cholesterol begins to coalesce into a fibrofatty plaque. As the plaque evolves, fibrosis develops and calcification may eventually appear. What later shows up as a coronary artery calcium score is therefore not the beginning of disease but evidence that the artery has been under biological stress for a long time.[3]
As plaque becomes unstable, rupture can occur. At this stage the body activates the clotting system to repair the injured vessel. Platelets aggregate and a protein called fibrin forms a mesh that stabilizes the clot. If that clot obstructs blood flow, the result may be a heart attack or stroke.
Understanding this sequence is essential. Cardiovascular disease is not simply a problem of cholesterol or clotting. It represents the culmination of metabolic dysfunction, inflammation, endothelial injury, plaque formation, and ultimately thrombosis.[1,2,16]
The Biological Stages of Atherosclerosis
The development of atherosclerosis follows a recognizable biological progression that has been well described in the cardiovascular literature.²¹⁶ Rather than appearing suddenly, the disease evolves through a sequence of biological events that unfold over many years. The process often begins with injury or dysfunction of the endothelium, the thin cellular layer that lines the inner surface of blood vessels. When the endothelium is exposed to metabolic stress, inflammatory signals, oxidative damage, or abnormal lipid particles, it becomes more permeable and more biologically active. This altered environment allows circulating immune cells and lipoproteins to enter the vessel wall.
As immune cells accumulate within the artery, macrophages begin to ingest retained lipoproteins, forming foam cells and creating what are known as fatty streaks, the earliest visible lesions of atherosclerosis. Over time, continued lipid accumulation and inflammatory signaling promote the growth of a fibrofatty plaque composed of cholesterol, immune cells, connective tissue, and smooth muscle cells. As the plaque enlarges, the artery may remodel outward to preserve blood flow, allowing the disease to progress silently for many years without causing symptoms.
Eventually, inflammatory processes within the plaque can weaken its structural integrity. When this occurs, the fibrous cap covering the plaque becomes vulnerable to rupture or erosion. Once the inner contents of the plaque are exposed to circulating blood, the body rapidly activates the clotting system. Platelets aggregate and fibrin forms a stabilizing mesh that creates a thrombus. If that thrombus obstructs blood flow within the artery, the result may be a heart attack or stroke.
Understanding these stages is critical because different therapies act at different points along this biological timeline. Understanding where therapies act within this progression helps clarify their role in cardiovascular prevention. Some treatments target metabolic dysfunction and inflammation early in the disease, while others act later by stabilizing plaque, preventing platelet aggregation, or dissolving fibrin within a clot.

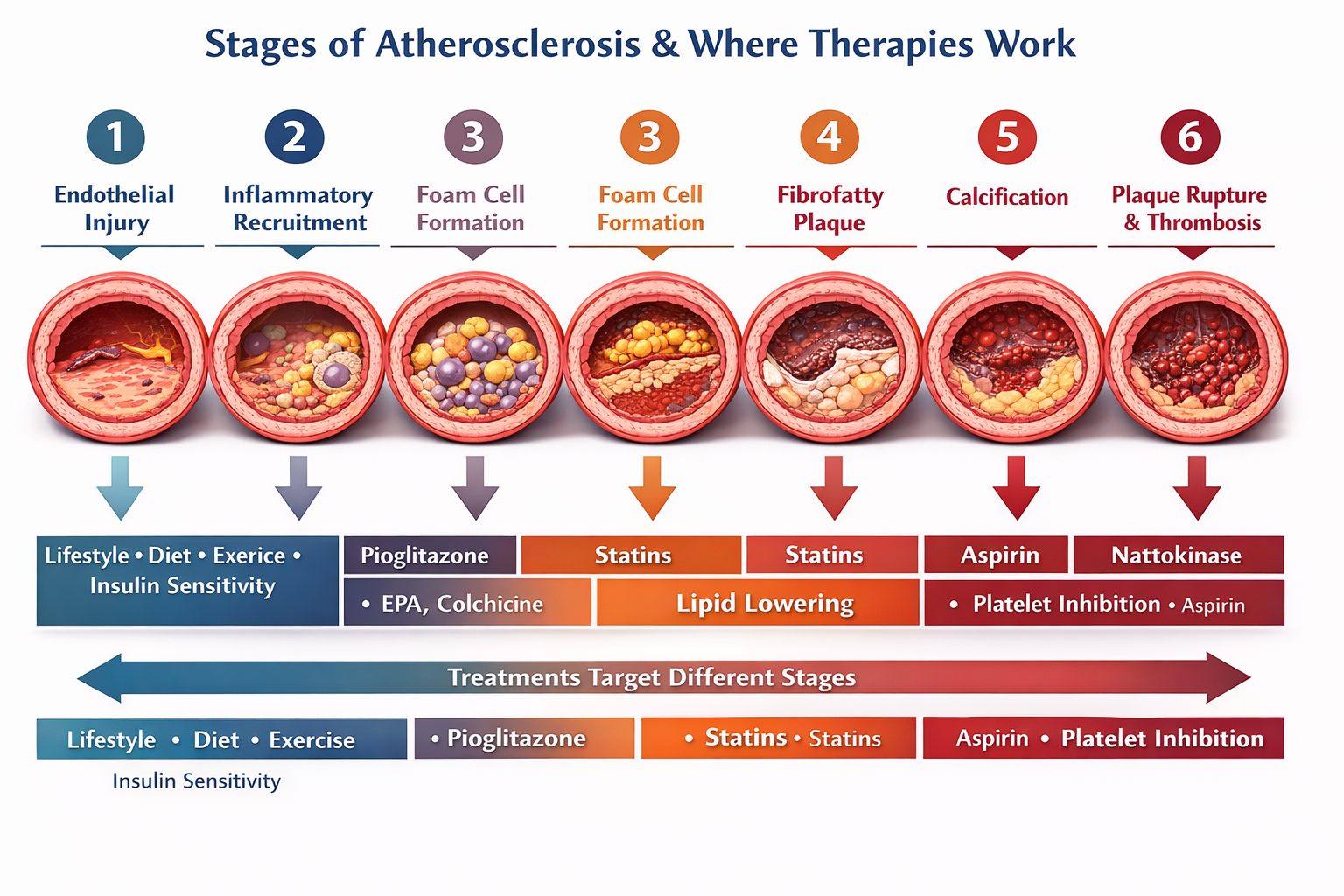
Figure 1: Stages of Atherosclerosis with Intervention Strategies
Stage 1 – Endothelial Injury- Metabolic dysfunction, oxidative stress, and inflammatory signaling damage the endothelial lining of the artery. Stage 2 – Immune Activation- Monocytes enter the vessel wall and differentiate into macrophages. Stage 3 – Foam Cell Formation- Macrophages ingest retained lipoproteins and become foam cells, forming fatty streaks. Stage 4 – Fibrofatty Plaque Formation- Cholesterol accumulates and a lipid core forms beneath a fibrous cap. Stage 5 – Plaque Instability- Inflammatory processes weaken plaque structure and activate platelets. Stage 6 – Thrombosis- Fibrin forms a mesh that stabilizes a clot, which may obstruct blood flow. This biological sequence illustrates why atherosclerosis is a long, progressive disease, and why therapies must target different stages of the process.
Aspirin and Platelet Biology
Aspirin has been one of the most widely used cardiovascular medications for decades. Its primary mechanism involves inhibition of platelet aggregation. Platelets are small circulating blood cells that respond rapidly to vascular injury. When a plaque ruptures, platelets adhere to the exposed surface of the artery and begin to aggregate, forming the initial plug that starts the clotting process.
Aspirin works by irreversibly inhibiting the cyclooxygenase-1 (COX-1) enzyme in platelets, preventing the formation of thromboxane A₂, a signaling molecule that promotes platelet activation and aggregation. By reducing thromboxane production, aspirin makes platelets less likely to clump together. This effect explains why aspirin has demonstrated clear benefit in secondary prevention, particularly in patients who have already experienced myocardial infarction, stroke, or established coronary artery disease.[15]
However, aspirin does not dissolve existing clots and does not directly affect fibrin metabolism. Its primary action occurs earlier in the clotting cascade, at the stage of platelet activation. For this reason, aspirin acts at a different stage of disease than nattokinase. While aspirin reduces platelet aggregation, nattokinase influences fibrin breakdown within the clot itself.
Natto itself has been consumed in Japan for centuries as a fermented soybean food and has been associated with favorable cardiovascular outcomes in several observational population studies.[5]
What Does Proteolytic Mean?
Nattokinase belongs to a class of enzymes known as proteolytic enzymes. The term proteolytic simply means capable of breaking down proteins. Proteins are long chains of amino acids that form structural and functional components throughout the body. Certain enzymes can cut these protein chains apart through a process known as proteolysis.
Nattokinase is a protease enzyme that breaks down fibrin, the structural protein that forms the mesh of a blood clot.[4] When a clot forms, platelets first aggregate to create the initial plug. Shortly afterward, fibrin strands form a network that stabilizes the clot.
In simple terms:
Platelets form the bricks of the clot.
Fibrin forms the mortar that stabilizes those bricks.
Nattokinase acts like a proteolytic blade that cuts through the fibrin mortar, weakening the clot and promoting fibrinolysis.
However, fibrin formation occurs very late in the atherosclerotic process, typically after plaque instability or rupture has already occurred.
.
Evidence for Nattokinase
Nattokinase is derived from natto, a traditional Japanese food made from fermented soybeans. The enzyme was first identified by Sumi and colleagues in 1987 when they observed that natto extracts possessed strong fibrin-dissolving properties.[4] Since that discovery, numerous laboratory studies have examined its biological effects.
Experimental research demonstrates that nattokinase can:
·directly degrade fibrin
·enhance endogenous fibrinolytic activity
·increase tissue plasminogen activator activity
·reduce plasminogen activator inhibitor-1
·influence markers of clot breakdown[5]
Animal studies have shown accelerated clot dissolution and reductions in fibrin accumulation after administration of nattokinase.[6] Human studies have also reported biological effects including increased fibrinolytic activity, reductions in fibrinogen levels, modest improvements in blood pressure, changes in coagulation parameters, and improved blood rheology.[5,17,18] Some small clinical investigations have also suggested improvements in carotid plaque measurements and lipid markers following supplementation.[7]
These findings indicate that nattokinase has measurable biological activity within the clotting system, particularly within the fibrinolytic pathway. However, most available studies involve relatively small populations and short follow-up periods. Large randomized trials evaluating major cardiovascular outcomes remain limited. For this reason, nattokinase should be viewed as a biologically active compound that influences fibrin and clot biology and may serve as an adjunct to—rather than a replacement for—established cardiovascular therapies targeting lipid metabolism, inflammation, and platelet activation.[5]
Table 2. Mechanistic Comparison of Nattokinase, Aspirin, and Statins


Why Fibrin Is the Final Event in Atherosclerotic Biology
Fibrin does not appear at the beginning of atherosclerosis. It appears at the end of a long biological story. Before fibrin is laid down, the artery has usually already passed through years of endothelial injury, immune activation, foam-cell formation, plaque growth, and often calcific remodeling. Only when plaque rupture or erosion exposes thrombogenic material to circulating blood does the coagulation cascade accelerate and fibrin become biologically dominant. In that sense, fibrin is not the origin of atherosclerosis but the structural signature of its most dangerous late complication. This is why nattokinase, while mechanistically interesting and potentially useful in selected situations, must be understood as acting on a final downstream event rather than on the upstream metabolic and inflammatory biology that created the vulnerable plaque in the first place.[1,2,21,22]
Biomarkers and the Biology of Clotting
Common Blood Tests That Help Reveal Cardiovascular Risk
A common question when discussing therapies such as aspirin or nattokinase is whether a specific blood test can determine which therapy should be used. The reality is that no single laboratory test can provide that answer. Cardiovascular disease involves multiple interacting systems including metabolism, inflammation, vascular biology, and coagulation. Because of this complexity, decisions about therapies that influence clotting biology must rely on a broader understanding of the patient’s biology.
Several biomarkers can help provide insight into this environment.
Fibrinogen
Fibrinogen is the precursor protein that is converted into fibrin during the clotting process. Elevated fibrinogen levels are associated with increased clot formation, higher cardiovascular risk, increased blood viscosity, and systemic inflammation. Numerous epidemiologic studies identify fibrinogen as an independent predictor of cardiovascular events.[8] From the perspective of nattokinase biology, fibrinogen represents the substrate that ultimately becomes fibrin. Elevated fibrinogen levels may therefore suggest that fibrin metabolism is contributing to vascular risk.
High-Sensitivity C-Reactive Protein (hs-CRP)
hs-CRP is one of the best validated markers of vascular inflammation. Elevated CRP levels reflect low-grade systemic inflammation, endothelial dysfunction, and increased cardiovascular risk. Many individuals with insulin resistance exhibit elevated CRP levels long before overt cardiovascular disease becomes apparent.[9]
Myeloperoxidase (MPO)
MPO is an enzyme released by activated white blood cells within inflamed plaques. Elevated MPO levels are associated with plaque vulnerability, oxidative stress, and increased cardiovascular risk.[10]
Lipoprotein-Associated Phospholipase A2 (LP-PLA2)
LP-PLA2 reflects inflammatory activity within atherosclerotic plaque and has been associated with plaque instability and cardiovascular risk.[11]
D-Dimer
D-dimer represents fibrin degradation products circulating in the bloodstream. Elevated levels indicate that clot formation and clot breakdown are occurring somewhere in the body, although the test is not specific to cardiovascular disease.[12]
The Importance of Arterial Imaging
At CardioCore, one of the most important diagnostic tools is not a blood test but arterial imaging. A coronary artery calcium scan (CAC) or coronary CT angiogram (CCTA) can determine whether atherosclerotic plaque is actually present in the coronary arteries. This information dramatically changes cardiovascular risk assessment because it allows physicians to visualize the disease itself rather than simply infer risk from laboratory markers or symptoms.
Blood tests can provide clues about inflammation, metabolism, or clotting biology, but they cannot directly show whether plaque is already forming within the arterial wall. Imaging, in contrast, allows clinicians to identify the structural presence of atherosclerosis. A calcium score measures calcified plaque burden, while coronary CT angiography can detect both calcified and non-calcified plaque and provide insight into plaque distribution and morphology. These findings help determine not only whether disease is present, but also how advanced the disease may be.[3,16]
The presence or absence of plaque significantly influences clinical decision-making. If no plaque is present, the risks of aggressive therapies such as blood-thinning medications may outweigh potential benefits. If plaque is present—particularly if it is extensive or biologically active—therapies that influence lipid biology, inflammation, platelet activation, or fibrin metabolism may become more relevant. Imaging therefore helps determine where a patient sits along the biological timeline of atherosclerosis.[3]
This approach differs significantly from traditional cardiovascular testing strategies that rely heavily on exercise or nuclear stress testing. Stress tests evaluate whether blood flow through the coronary arteries becomes restricted during physical exertion. While this method can detect severe flow-limiting blockages, stress testing does not identify the presence of early atherosclerosis, nor does it quantify total plaque burden or provide meaningful insight into plaque biology.[23]
In other words, stress testing does not diagnose the disease itself. It only detects whether a blockage has become severe enough to restrict blood flow. Many myocardial infarctions occur in arteries that previously had only moderate narrowing, meaning that a stress test can appear normal even when substantial plaque is already present within the arterial wall.[21]
Another limitation of stress testing is the phenomenon known as plaque–stenosis discordance. Stenosis refers to the degree of narrowing within the artery, while plaque burden refers to the total amount of atherosclerotic disease within the vessel wall. These two measurements do not always correlate. An artery may contain a large amount of plaque that expands outward within the vessel wall—a process known as positive remodeling—while producing little or no luminal narrowing. In these situations, stress testing may appear normal even though significant atherosclerotic disease is present.
Coronary CT angiography is uniquely capable of identifying this discordance. By visualizing plaque within the vessel wall rather than only measuring luminal narrowing, arterial imaging can detect disease at an earlier stage and reveal biologically active plaque that may not yet produce flow limitation.[21,24]
Radiation exposure is another important consideration. Nuclear stress testing often exposes patients to substantially higher radiation doses than modern coronary calcium scanning or contemporary coronary CT angiography protocols. Yet despite this higher radiation exposure, stress testing frequently provides less information about the underlying biology of atherosclerosis.[23]
For these reasons, arterial imaging has become an increasingly important tool in modern preventive cardiology. Rather than waiting for a blockage to restrict blood flow, imaging allows physicians to identify atherosclerosis earlier, measure plaque burden more accurately, and align therapy with the biological stage of disease.
In the context of therapies such as aspirin or nattokinase, this information becomes especially valuable. If plaque is absent, clot-focused therapies may provide little benefit. If plaque is present and biologically active, therapies targeting inflammation, platelet activity, lipid metabolism, or fibrin biology may become more appropriate.
Arterial imaging therefore shifts cardiovascular medicine away from reacting to late-stage complications and toward understanding and managing the disease process itself.
Where Different Therapies Act in the Disease Process
Different cardiovascular therapies act at different points along the biological timeline of atherosclerosis. Understanding where these therapies work helps clarify why no single treatment can address the entire disease process.
Some therapies act very early, before plaque even begins to form. At this stage the primary drivers of disease are metabolic dysfunction, insulin resistance, oxidative stress, and inflammation affecting the endothelium. Medications such as pioglitazone work at this early level by improving insulin sensitivity and reducing inflammatory signaling through activation of the PPAR-γ pathway.[13] By improving metabolic regulation and lowering inflammatory stress within the vascular system, therapies like pioglitazone may help stabilize the biological environment that leads to endothelial injury.
Icosapent ethyl (EPA) also acts relatively early in the disease process. EPA exerts anti-inflammatory effects, reduces oxidative stress, and improves endothelial signaling. It has also been shown to influence plaque stability and reduce cardiovascular events in large clinical trials.[14] By improving vascular biology and reducing inflammatory stress within the arterial wall, EPA may help slow the processes that allow plaque to develop and evolve.
As atherosclerosis progresses, lipid accumulation becomes a dominant driver of plaque growth. At this stage, statins play a central role. Statins reduce circulating ApoB-containing lipoproteins and lower LDL cholesterol, thereby decreasing the lipid particles that penetrate the arterial wall. Beyond lipid lowering, statins also exert anti-inflammatory effects and promote stabilization of atherosclerotic plaques. For this reason, statins occupy a critical middle position in the biological progression of atherosclerosis, acting during the stages of foam-cell accumulation and fibrofatty plaque development.[20]
Later in the disease process, the biological focus shifts toward plaque instability and thrombosis. When plaques become inflamed or structurally weakened, platelets are activated and begin forming the initial clot. Aspirin works at this stage by inhibiting platelet activation and preventing platelet aggregation, thereby reducing the likelihood that a clot will rapidly expand and obstruct blood flow.
Finally, at the very end of the cascade, the clotting system produces fibrin, a structural protein that forms the mesh stabilizing the clot. This is the stage where nattokinase exerts its primary biological effect. As a proteolytic enzyme capable of degrading fibrin, nattokinase acts within the fibrinolytic system to help break down the fibrin scaffold of a clot.
Seen within this broader biological framework, it becomes clear that different therapies target different stages of cardiovascular disease. Some act early by addressing metabolic dysfunction and inflammation, others slow plaque growth and stabilize arterial biology, while still others influence platelet activity or fibrin clot formation.
Understanding where these therapies operate along the timeline of disease helps explain why effective cardiovascular prevention often requires multiple complementary strategies rather than a single intervention.

Fire, Fuel, and Sparks
At CardioCore, we often describe cardiovascular disease using a simple analogy that helps illustrate how the disease develops over time.
Inflammation represents the fire within the arteries.
This inflammatory environment is driven by immune activation, oxidative stress, and metabolic disturbances that injure the vascular lining and destabilize plaque.
Metabolic dysfunction—particularly insulin resistance—serves as the fuel feeding that fire.
When metabolism becomes dysregulated, excess glucose, abnormal lipoproteins, oxidative stress, and hormonal signaling disturbances create a biological environment that continuously feeds vascular inflammation.
Thrombosis represents the spark produced by this unstable environment.
When plaque becomes inflamed or structurally weakened, the body activates the clotting system in an attempt to repair the injured vessel. Platelets aggregate, fibrin forms, and a thrombus may develop.
Nattokinase may help control some of these sparks by dissolving fibrin within the clot. But removing sparks alone does not extinguish the fire. If the underlying metabolic and inflammatory drivers remain active, the conditions that generate plaque instability and thrombosis continue to exist.
In this sense, thrombosis is often the final event of a much longer biological process. Addressing clot formation without addressing the forces that produced the plaque is similar to extinguishing sparks while leaving the fire burning.
True cardiovascular prevention therefore requires identifying and treating the underlying biological conditions that fuel vascular inflammation in the first place.
Why Stage-Based Medicine Matters
Understanding cardiovascular disease through a stage-based framework fundamentally changes how we approach treatment and prevention.
Atherosclerosis is not a single event but a progressive biological process that evolves over decades. Early in the disease, metabolic dysfunction, insulin resistance, and inflammatory signaling injure the endothelium and initiate plaque formation. As the disease advances, lipid accumulation and immune activation drive plaque growth and structural remodeling of the arterial wall. In later stages, plaque instability activates platelets and the clotting cascade, ultimately leading to thrombosis.
Because different biological forces dominate at different points in this progression, therapies that target one stage of disease cannot address the entire process.
Some treatments work early by improving metabolic health and reducing inflammatory signaling. Others slow plaque development by lowering ApoB-containing lipoproteins and stabilizing arterial biology. Still others act later by reducing platelet activation or dissolving fibrin within a clot.
When therapies are aligned with the stage of disease, treatment becomes more precise and biologically rational. Rather than applying a single intervention to every patient, physicians can tailor therapy to the dominant biological processes occurring within the arteries.
Stage-based medicine therefore represents a shift away from a one-size-fits-all approach toward a more integrated and personalized model of cardiovascular prevention.
The Final Perspective
Nattokinase is an intriguing natural compound with measurable biological effects on fibrin metabolism and clot biology. Laboratory studies and early clinical investigations demonstrate that it can enhance fibrinolytic activity and influence pathways involved in thrombus formation.
But to understand the true role of nattokinase, it must be placed within the broader biological framework of atherosclerosis.
Fibrin formation represents one of the final stages of cardiovascular disease, not the beginning. By the time fibrin stabilizes a clot, the artery has often already experienced years—or decades—of metabolic stress, inflammatory signaling, lipid accumulation, and plaque development.
For this reason, the most powerful strategy in cardiovascular medicine is not simply dissolving clots after they form. It is identifying and correcting the biological forces that make those clots possible.
True cardiovascular prevention requires addressing the metabolic terrain that shapes arterial health. This includes recognizing the roles of insulin resistance, chronic inflammation, endothelial dysfunction, oxidative stress, and abnormal lipid biology in driving plaque formation and instability.
At CardioCore, the goal is not simply to react to the final event. The goal is to understand why the body becomes vulnerable to that event in the first place.
Because the most effective strategy in cardiovascular medicine is not responding to the spark of thrombosis after it occurs.
It is preventing the fire and fuel that allow that spark to ignite.
“True cardiovascular prevention begins not with dissolving
the clot, but with understanding the biology that allowed
the clot to form”…CardioCore Metabolic Wellness Center
Book a Call and Find Out More... Dr John
CLICK HERE TO BOOK A DISCOVERY CALL
CLICK HERE TO JOIN OUR PRIVATE CardioCore COMMUNITY
CLICK HERE TO SPEAK TO OUR VIRTUAL ASSISTANT
The information presented here is intended for educational purposes only. Patients should not start, stop, or change any medications without first discussing these decisions with their treating physician.
References
1.Libby P. Inflammation in atherosclerosis. Nature. 2002;420:868-874.
2.Ross R. Atherosclerosis—an inflammatory disease. N Engl J Med. 1999;340:115-126.
3.Greenland P, Blaha MJ, Budoff MJ, Erbel R, Watson KE. Coronary calcium scoring and cardiovascular risk assessment. Circulation. 2018.
4.Sumi H, Hamada H, Tsushima H, Mihara H, Muraki H. A novel fibrinolytic enzyme (nattokinase) in natto. Experientia. 1987;43:1110-1111.
5.Chen H, McGowan EM, Ren N, et al. Nattokinase: a promising alternative in prevention and treatment of cardiovascular disease. Integr Med Res. 2018;7:24-28.
6.Fujita M, Nomura K, Hong K, Ito Y, Asada A, Nishimuro S. Thrombolytic effect of nattokinase on chemically induced thrombosis. Biol Pharm Bull. 1995;18:1387-1391.
7.Ren N, Chen H, Li Y, et al. Nattokinase supplementation and carotid artery atherosclerosis progression. Scientific Reports. 2022.
8.Danesh J, Lewington S, Thompson SG, et al. Plasma fibrinogen level and cardiovascular risk. JAMA. 2005;294:1799-1809.
9.Ridker PM. C-reactive protein and prediction of cardiovascular events. N Engl J Med. 2007;356:2139-2141.
10.Brennan ML, Penn MS, Van Lente F, et al. Prognostic value of myeloperoxidase in patients with chest pain. N Engl J Med. 2003;349:1595-1604.
11.Ballantyne CM, Hoogeveen RC, Bang H, et al. Lipoprotein-associated phospholipase A2 and coronary risk. Circulation. 2004;109:837-842.
12.Adam SS, Key NS, Greenberg CS. D-dimer antigen: current concepts and future prospects. Blood. 2009;113:2878-2887.
13.Dormandy JA, Charbonnel B, Eckland DJA, et al. Secondary prevention of macrovascular events in type 2 diabetes (PROactive Study). Lancet. 2005;366:1279-1289.
14.Bhatt DL, Steg PG, Miller M, et al. Cardiovascular risk reduction with icosapent ethyl (REDUCE-IT). N Engl J Med. 2019;380:11-22.
15.Antithrombotic Trialists’ Collaboration. Aspirin in primary and secondary prevention of vascular disease. Lancet. 2009;373:1849-1860.
16.Libby P, Buring JE, Badimon L, et al. Atherosclerosis. Nat Rev Dis Primers. 2019;5:56.
17.Hsia C-H, Shen M-C, Lin J-S, et al. Nattokinase decreases plasma fibrinogen, factor VII, and factor VIII. Nutr Res. 2009;29:190-196.
18.Jensen GS, Lenninger M, Ero MP, Benson KF. Nattokinase consumption associated with reduced blood pressure. Hypertens Res. 2016;39:217-223.
19.Ridker PM, Everett BM, Thuren T, et al. Antiinflammatory therapy with canakinumab (CANTOS). N Engl J Med. 2017;377:1119-1131.
20.Nissen SE, Nicholls SJ, Sipahi I, et al. Statin therapy and regression of coronary atherosclerosis (ASTEROID). JAMA. 2006;295:1556-1565.
21.Virmani R, Burke AP, Farb A, Kolodgie FD. Pathology of the vulnerable plaque. J Am Coll Cardiol. 2006;47:C13-C18.
22.Libby P, Pasterkamp G. Requiem for the vulnerable plaque. Eur Heart J. 2015;36:2984-2987.
23.Nicholls SJ et al. Intensive statin therapy and coronary plaque regression (SATURN). Lancet. 2011.
24.Douglas PS, Hoffmann U, Patel MR, et al. Outcomes of anatomical versus functional testing for coronary artery disease (PROMISE Trial). N Engl J Med. 2015;372:1291-1300.
25.Sciales J. The Lumen Is Not the Disease: Plaque Biology and the Burden–Stenosis Discordance in Coronary Artery Disease. Unpublished manuscript.
