
Hidden in Plain Sight: “Euglycemic Diabetes” as the Earliest Signal of Cardiometabolic Disease
Hidden in Plain Sight: “Euglycemic Diabetes” as the Earliest Signal of Cardiometabolic Disease
Normal Glucose Does Not Mean Normal Metabolism &
Heart Disease Does Not Occur Without Insulin Resistance
Dr John ScialesDirector: CardioCore Metabolic Wellness Center
Introduction
Traditional assessment of glucose metabolism relies heavily on static glycemic markers such as fasting glucose and hemoglobin A1c. While useful for identifying overt dysglycemia, these measures fail to capture the dynamic physiology underlying metabolic regulation. In particular, they do not reflect the magnitude of insulin secretion required to maintain apparently normal glucose levels. As a result, significant metabolic dysfunction, most notably insulin resistance, can remain undetected for years, masked by compensatory hyperinsulinemia [6,7,21].
This limitation is not theoretical. It is clinically observable in the very population we are trying to protect. In patients with established coronary artery disease, metabolic dysfunction is frequently present long before glucose abnormalities emerge. In a pivotal study by Jesús et al., individuals with coronary disease and no prior diagnosis of diabetes underwent oral glucose tolerance testing. Despite “normal” fasting glucose and hemoglobin A1c, nearly two-thirds demonstrated abnormal glucose tolerance, and within three years, up to 88% progressed to prediabetes or overt diabetes [1].
Importantly, insulin levels were not measured in that study. Had insulin dynamics been assessed, the true prevalence of metabolic dysfunction would almost certainly have been even higher. What this reveals is a fundamental truth: cardiovascular disease does not begin when glucose becomes abnormal. It begins earlier, during a phase of compensated insulin resistance in which glucose remains normal only because insulin is elevated.
This state can be understood as “Euglycemic Diabetes”, a term that is intentionally paradoxical. “Euglycemic” means normal glucose, and when paired with “diabetes,” it forces a reconsideration of what diabetes truly represents. The condition is not absent, it is simply hidden. We are not missing the disease because it is not there; we are missing it because we are not looking in the right way.
The problem is not that the disease is silent, the problem is that our tools are.
Discordance Between Glucose and Insulin: The Illusion of Normalcy
The glucose response in metabolic testing often appears, at first glance, to be within normal limits, with a modest rise followed by a return toward baseline. By conventional diagnostic criteria, this would be interpreted as normal glucose tolerance. However, when evaluated alongside insulin, a markedly different and clinically significant picture emerges.
The apparent normality of glucose is achieved only through a disproportionately elevated and prolonged insulin response. At baseline, insulin levels are often already elevated, reflecting underlying insulin resistance. Following glucose ingestion, insulin rises sharply, peaking at levels far exceeding what would be expected for the degree of glycemic excursion. This reflects reduced insulin sensitivity, requiring the pancreas to secrete substantially greater amounts of insulin to maintain euglycemia. In this context, glucose values do not represent metabolic health, but rather successful physiologic compensation [7].
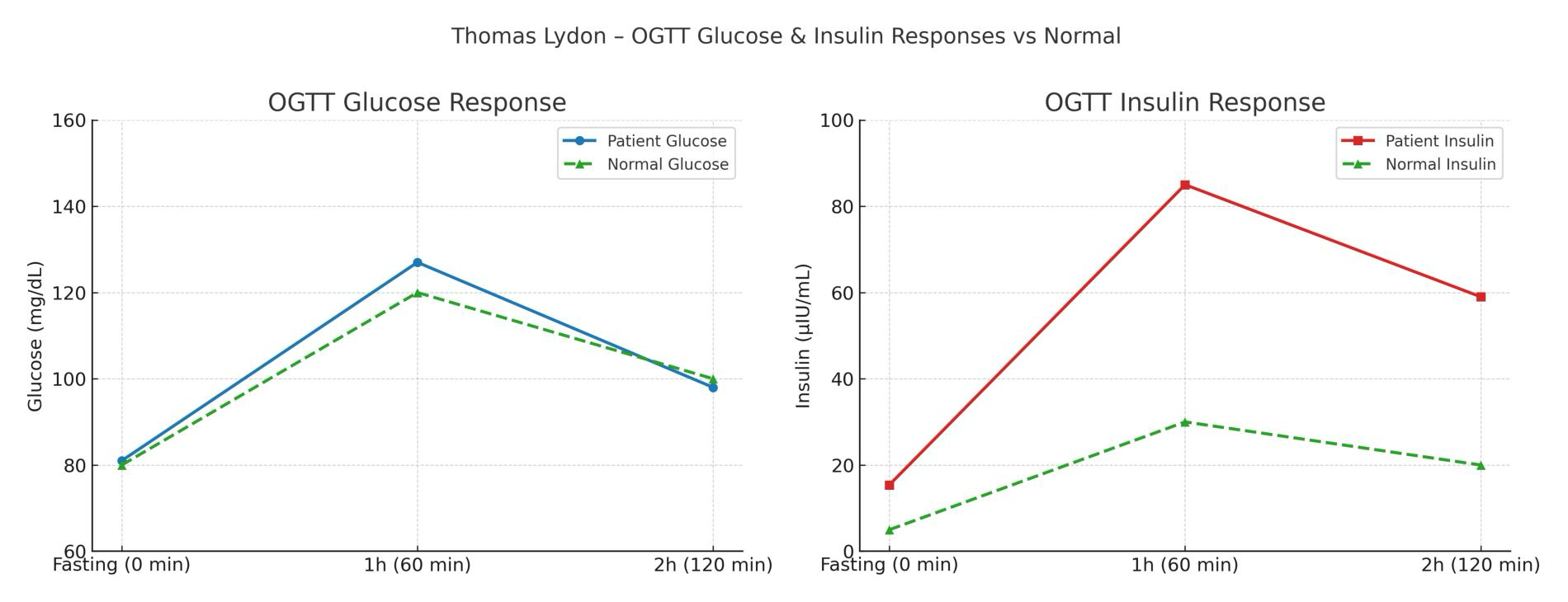
To illustrate this clearly, consider a representative oral glucose tolerance test with insulin measurements from a 64-year-old patient with established premature cardiovascular disease and a hemoglobin A1c of 5.1%, a value that, by conventional standards, would suggest metabolic health. Fasting glucose is approximately 85 mg/dL, rising modestly to approximately 122 mg/dL at one hour and returning to approximately 98 mg/dL by two hours. By conventional criteria, this would be interpreted as normal glucose tolerance. However, the insulin response tells a fundamentally different story. Fasting insulin is elevated at approximately 15–20 µIU/mL, rises sharply to approximately 80–85 µIU/mL at one hour, and remains abnormally elevated at approximately 55–60 µIU/mL at two hours, rather than returning toward baseline.
This is not normal physiology. This is a system under strain. It is a system maintaining normal glucose at the cost of excessive insulin exposure.Illustration: Normal glucose curves can mask significant metabolic dysfunction. Elevated and prolonged insulin responses reveal underlying insulin resistance and early cardiometabolic disease despite “normal” laboratory values.
Temporal Mismatch and Metabolic Inefficiency
As the test progresses, glucose levels normalize relatively quickly, yet insulin remains elevated well beyond the period of glucose absorption. In a metabolically healthy individual, insulin secretion closely parallels glucose excursions, rising and falling in synchrony. Here, the persistence of elevated insulin despite normalized glucose reflects ongoing compensatory secretion and impaired metabolic efficiency, particularly at the level of skeletal muscle, the primary site of insulin-mediated glucose disposal [22].
This temporal dissociation between glucose and insulin dynamics represents a hallmark of insulin resistance. It signifies a system in which greater hormonal input is required to achieve a diminishing metabolic effect, consistent with progressive loss of insulin sensitivity at the cellular level [6,21].
Reactive Hypoglycemia: The Consequence of Insulin Overshoot
The late phase of the test further exposes this dysfunction. Glucose may decline to levels consistent with reactive hypoglycemia, often in the range of approximately 55–60 mg/dL. This finding is not attributable to inadequate glucose availability, but rather to excessive insulin activity.
Continued insulin-mediated glucose uptake in the absence of corresponding glucose input results in a physiologic overshoot and subsequent decline in circulating glucose levels [25]. This triggers counter-regulatory responses, including activation of the sympathetic nervous system, leading to symptoms such as hunger, fatigue, irritability, and a drive for rapidly absorbable carbohydrates.
Over time, this cycle promotes increased caloric intake, weight gain, and further deterioration of insulin sensitivity, reinforcing a self-perpetuating loop of metabolic dysfunction [20,24].
Hyperinsulinemia as a Driver of Vascular Disease
From a pathophysiologic perspective, hyperinsulinemia is not a benign or adaptive state. It is biologically active and exerts direct effects on the vasculature.
Elevated insulin levels contribute to endothelial dysfunction, increased oxidative stress, and activation of pro-inflammatory signaling pathways, including NF-κB, which play a central role in the initiation and progression of atherosclerosis [5,21–23]. Insulin also promotes vascular smooth muscle proliferation, enhances sympathetic tone, and contributes to sodium retention and hypertension.
In the setting of established or suspected coronary artery disease, this metabolic profile reflects ongoing disease activity rather than metabolic stability. It represents a form of residual cardiovascular risk that is not captured by traditional markers such as glucose, hemoglobin A1c, or even LDL cholesterol.
Clinical Implications: Beyond Glucose-Centric Thinking
Accordingly, patients with this metabolic profile should not be considered metabolically normal, but rather metabolically compensated, maintaining normal glucose at the expense of excessive insulin secretion.
This distinction is fundamental. It shifts the clinical focus from downstream markers, such as glucose, to upstream pathophysiology, including insulin resistance and inflammation. The insulin curve represents the true signal of disease, revealing elevated baseline levels, exaggerated peak response, prolonged elevation, and delayed normalization.
In patients with cardiovascular disease, the presence of normal glucose should not reassure the clinician. Instead, it should prompt further investigation. The data from coronary populations clearly demonstrate that “normal” glucose frequently masks underlying metabolic dysfunction [1].
The clinical implication is clear: any patient with cardiovascular disease and normal glycemic markers should undergo dynamic metabolic testing, including oral glucose tolerance testing with insulin measurements.
While OGTT improves detection of dysglycemia compared to fasting glucose and hemoglobin A1c, it remains a fundamentally glucose-centered assessment. The earliest pathophysiologic disturbance, hyperinsulinemia, often precedes measurable glucose abnormalities, suggesting that even OGTT underestimates the true burden of cardiometabolic disease[26].
“Euglycemic Diabetes”: Defining the Hidden Cardiometabolic State
The findings presented here support the recognition of a distinct and clinically meaningful condition: “Euglycemic Diabetes”.
This state is defined by normal glucose levels in the presence of pathologic insulin dynamics. It represents a phase of cardiometabolic disease in which insulin resistance and compensatory hyperinsulinemia are already present and actively driving vascular injury, despite the absence of abnormal glycemic markers.
In practical terms, these patients are not metabolically normal. They are metabolically compensated.
The distinction is critical. Traditional definitions of diabetes rely on glucose thresholds. However, by the time glucose becomes abnormal, the underlying disease process has often been present for years. During that time, hyperinsulinemia has already exerted its effects on endothelial function, inflammatory signaling, and plaque development.
Euglycemic diabetes therefore represents an earlier stage of the same disease continuum, one that is largely invisible to conventional testing but highly relevant to cardiovascular risk.
“Accordingly, in patients with cardiovascular disease and normal glycemic markers, euglycemic diabetes should be considered the default metabolic state unless proven otherwise.”
Identifying Euglycemic Diabetes in Clinical Practice
If this condition exists, the next question is straightforward: how do we identify it?
Even when oral glucose tolerance testing is performed, reliance on glucose values alone may fail to detect the earliest stages of metabolic dysfunction. Large coronary artery disease cohorts have demonstrated that post-load glucose improves detection of dysglycemia compared to fasting glucose and hemoglobin A1c, yet remains limited to glucose-based assessment and does not capture underlying insulin resistance[26].
The answer is equally straightforward, yet rarely implemented.
In any patient with established or suspected cardiovascular disease, particularly those with “normal” glucose and hemoglobin A1c, further metabolic evaluation is not optional—it is necessary.
This begins with dynamic testing. A standard oral glucose tolerance test must be paired with insulin measurements, including fasting, 30-minute, 60-minute, and 120-minute values. It is the insulin curve, not the glucose curve, that reveals the underlying physiology.
Patterns consistent with euglycemic diabetes include elevated fasting insulin, exaggerated early-phase insulin response, prolonged elevation beyond glucose normalization, and delayed return to baseline. In some cases, a late glucose nadir reflecting reactive hypoglycemia further supports the diagnosis.
Beyond dynamic testing, evaluation must extend to the broader cardiometabolic environment. Insulin resistance does not occur in isolation. It is influenced by hormonal regulation, inflammatory signaling, mitochondrial function, and the gut microbiome.
Accordingly, assessment should include markers of inflammation, metabolic flexibility, and hormonal balance, including cortisol and sex hormones. In parallel, structural assessment with coronary imaging provides confirmation of disease burden and plaque phenotype, linking metabolic dysfunction to its vascular consequence.
Taken together, this approach allows for identification of disease at a stage when it is still biologically active, but not yet clinically obvious.
Therapeutic Implications: Moving Beyond Glucose Toward Insulin Resistance and Inflammatory Biology
In this context, therapeutic decision-making must be anchored in the dominant pathophysiology, which is not hyperglycemia but insulin resistance with compensatory hyperinsulinemia and associated inflammatory vascular signaling.
Plasma glucose is normal. It is not the disease, and therefore not the primary therapeutic target. The metabolic disturbance is defined by excessive insulin exposure, which drives endothelial dysfunction, oxidative stress, and activation of inflammatory pathways, including NF-κB [5,7,21–23]. A glucose-centric approach risks treating a surrogate marker rather than the underlying disease process.
Upstream First: Reframing Therapeutic Strategy in Cardiometabolic Disease
Before pharmacologic therapy is considered, a more fundamental question must be addressed: what is driving the biology?
In the setting of euglycemic diabetes, the dominant abnormality is not hyperglycemia, but insulin resistance with compensatory hyperinsulinemia and an associated pro-inflammatory metabolic environment. Treating this condition by focusing solely on glucose is analogous to treating fever without identifying infection. The visible marker is not the disease; it is the consequence of it.
This distinction reframes therapeutic strategy. Rather than initiating treatment based on laboratory thresholds alone, the clinical objective becomes identification of upstream drivers, including metabolic inflexibility, chronic inflammatory signaling, hormonal dysregulation, and environmental inputs that influence energy utilization and storage. These variables determine whether the system remains in a state of compensation or progresses toward overt disease.
Accordingly, the initial phase of management should not be reflexively pharmacologic. It should be investigative. The goal is to define the terrain in which the disease developed, because it is within that terrain that the opportunity for meaningful intervention exists.
Functional Cardiometabolic Evaluation: Identifying the Drivers of Disease
A comprehensive therapeutic approach begins with characterization of the metabolic and inflammatory environment at a systems level.
This includes assessment of genetic predispositions that influence insulin signaling, lipid handling, inflammatory pathways, and vascular biology. It includes advanced metabolic profiling to determine substrate utilization, mitochondrial efficiency, and the presence of metabolic inflexibility, a state in which the body is unable to appropriately switch between glucose and fatty acid metabolism.
Hormonal regulation is central to this process. Dysregulation of cortisol, alterations in sex hormones such as testosterone, and broader endocrine imbalances directly influence insulin sensitivity, visceral adiposity, and inflammatory tone. These are not secondary findings; they are primary contributors to the metabolic phenotype.
Equally important is the gut microbiome, which functions as a key regulator of immune signaling, nutrient metabolism, and systemic inflammation. Disruption of microbial balance can promote endotoxemia, amplify inflammatory pathways, and exacerbate insulin resistance independent of caloric intake.
These biologic insights are then integrated with lifestyle and environmental factors, including nutritional patterns, degree of carbohydrate tolerance, physical activity, sleep quality, stress physiology, and behavioral influences. Nutrition is not simply caloric input; it is biologic information that directly modulates metabolic pathways and inflammatory signaling.
Targeted supplementation may be used strategically within this framework to support mitochondrial function, reduce oxidative stress, and modulate inflammatory pathways. However, these interventions are not applied empirically. They are selected based on the specific physiologic disturbances identified through comprehensive evaluation.
This approach does not replace pharmacologic therapy when indicated. It precedes it. It ensures that treatment is directed not only at the manifestations of disease, but at the mechanisms that sustain it.
The question is not which drug to start. The question is which variable to change.
From Upstream Drivers to Targeted Pharmacologic Intervention
While identification and modification of upstream drivers represent the foundation of cardiometabolic care, clinical reality often reflects a more advanced stage of disease. By the time cardiovascular disease is established, whether through imaging, symptoms, or prior clinical events, the biology is no longer confined to metabolic dysfunction alone. Structural and inflammatory consequences are already present within the vasculature.
At this stage, an exclusively upstream approach, while necessary, is not sufficient.
The presence of atherosclerotic plaque, endothelial dysfunction, and active inflammatory signaling requires concurrent downstream intervention. These therapies are, by definition, reactive. They address the expression of disease rather than its origin. However, when selected appropriately, they can alter disease trajectory, stabilize plaque, and reduce near-term cardiovascular risk.
The critical question is not whether pharmacologic therapy should be used, but how it is selected.
In a glucose-centric model, treatment is guided by laboratory thresholds, often leading to therapies that lower glucose without addressing the underlying metabolic disturbance. In contrast, a biology-driven approach aligns pharmacologic intervention with the dominant pathophysiology, insulin resistance, hyperinsulinemia, and inflammation.
This distinction reframes drug therapy from a protocol-based response to a targeted strategy. Medications are not chosen simply to normalize numbers, but to influence the mechanisms driving disease progression.
Accordingly, the following therapeutic considerations are presented not as isolated pharmacologic options, but as interventions that, to varying degrees, align with the upstream biology of cardiometabolic disease.
When disease is already present, the objective is not simply to treat it, but to treat it in a manner consistent with how it developed.
Pioglitazone: Targeting Insulin Resistance, Fuel Utilization, and Inflammatory Biology
Pioglitazone represents one of the most mechanistically aligned pharmacologic therapies in cardiometabolic disease, as it directly targets the upstream drivers of pathology—insulin resistance, hyperinsulinemia, disordered fuel utilization, and chronic inflammation. As a potent agonist of peroxisome proliferator–activated receptor gamma (PPAR-γ), pioglitazone exerts coordinated effects across skeletal muscle, adipose tissue, and liver, restoring metabolic balance at both the cellular and systemic level.
At the core of insulin resistance is a disruption in substrate utilization, often described through the framework of the Randle cycle [27]. In this state, elevated circulating free fatty acids (FFA), driven by adipose tissue insulin resistance, inhibit glucose oxidation within skeletal muscle. Increased fatty acid flux into mitochondria leads to accumulation of intermediates such as acetyl-CoA and citrate, which suppress key glycolytic enzymes and impair glucose uptake and utilization. The result is metabolic inflexibility: the inability of the system to appropriately switch between lipid and glucose oxidation based on physiologic demand.
Pioglitazone directly addresses this dysfunction.
Through PPAR-γ activation, it suppresses lipolysis in adipose tissue, reducing circulating FFA levels and thereby decreasing lipid oversupply to skeletal muscle and liver. This reduction in FFA flux relieves inhibition of glucose oxidation pathways, restoring insulin-mediated glucose uptake and improving mitochondrial efficiency. In effect, pioglitazone rebalances the competitive relationship between fatty acids and glucose, allowing for appropriate substrate switching and improved energy production.
This restoration of metabolic flexibility has downstream consequences that extend beyond glycemic control. By reducing ectopic lipid accumulation in skeletal muscle and hepatic tissue, pioglitazone alleviates lipotoxic stress, a key driver of mitochondrial dysfunction, oxidative stress, and impaired insulin signaling. Mitochondria are no longer forced into a state of chronic fatty acid overload, and cellular energy production becomes more efficient and less inflammatory.
Simultaneously, pioglitazone induces a fundamental shift in adipose tissue biology. It promotes differentiation of smaller, insulin-sensitive adipocytes and facilitates redistribution of lipid from visceral and ectopic depots into subcutaneous stores, which are metabolically less inflammatory [28]. This process is accompanied by a marked increase in adiponectin, a key adipokine that enhances fatty acid oxidation, improves insulin sensitivity, and exerts direct anti-inflammatory and anti-atherogenic effects at the vascular level [29].
Beyond metabolic regulation, pioglitazone exerts potent anti-inflammatory effects through inhibition of nuclear factor kappa B (NF-κB), a central transcription factor linking metabolic dysfunction to vascular disease. NF-κB activation drives expression of pro-inflammatory cytokines, adhesion molecules, and oxidative stress pathways that promote endothelial dysfunction and atherosclerotic plaque progression [5,21–23]. Through PPAR-γ–mediated suppression of NF-κB signaling, pioglitazone reduces vascular inflammation at its source, rather than simply attenuating downstream inflammatory markers.
These integrated effects—improved insulin sensitivity, restoration of fuel utilization, reduction in lipotoxicity, and suppression of inflammatory signaling—translate into measurable vascular benefit. Imaging studies such as PERISCOPE have demonstrated reduced progression of coronary atherosclerosis compared to sulfonylurea therapy, while CHICAGO showed improvements in carotid intima-media thickness. Outcome trials including PROactive and IRIS further support reductions in major adverse cardiovascular events, particularly in insulin-resistant populations [10].
From a cardiometabolic perspective, pioglitazone is not simply a glucose-lowering agent. It is a therapy that restores biologic balance within the metabolic system. It reduces the need for compensatory hyperinsulinemia, corrects disordered substrate utilization, improves mitochondrial efficiency, redistributes lipid away from toxic depots, enhances protective adipokine signaling, and suppresses inflammatory pathways central to vascular injury.
In this context, pioglitazone does not treat a laboratory value—it treats the biology that creates the disease.
Metformin: Mechanistic Misalignment in Euglycemic Disease
Metformin remains one of the most widely prescribed therapies in metabolic medicine; however, in the specific context of euglycemic diabetes—where the dominant abnormality is insulin resistance with compensatory hyperinsulinemia—its mechanism is fundamentally misaligned with the underlying biology.
Its primary action is the suppression of hepatic gluconeogenesis through inhibition of mitochondrial respiratory chain complex I, leading to activation of AMP-activated protein kinase (AMPK) and a subsequent reduction in hepatic glucose output [6]. While effective in lowering plasma glucose, this mechanism does not directly correct the core defect of insulin resistance at the level of skeletal muscle, nor does it meaningfully reduce compensatory hyperinsulinemia. In a state where glucose is already normal, lowering glucose further does not address the disease—it bypasses it.
From a bioenergetic perspective, metformin induces a mild state of mitochondrial stress. By inhibiting oxidative phosphorylation, it reduces ATP production and shifts cellular metabolism toward increased glycolysis. While this may be beneficial in reducing hepatic glucose production, it comes at the cost of reduced mitochondrial efficiency, particularly in tissues with high energy demand such as skeletal muscle and myocardium [30].
This mitochondrial effect may also interfere with physiologic adaptation to exercise. Exercise-induced improvements in insulin sensitivity and metabolic flexibility are mediated, in part, through mitochondrial biogenesis and enhanced oxidative capacity. Metformin has been shown to blunt these adaptive responses, potentially attenuating one of the most powerful non-pharmacologic interventions available for improving insulin sensitivity [31].
In addition, metformin does not meaningfully correct disordered fuel utilization. It does not reduce elevated free fatty acid flux from adipose tissue, does not restore substrate switching at the level of skeletal muscle, and does not address lipotoxicity within muscle or liver. The Randle cycle remains dysregulated, and metabolic inflexibility persists [27].
Its effects on inflammation are indirect and modest. While AMPK activation may exert secondary anti-inflammatory effects, metformin does not directly inhibit central inflammatory pathways such as NF-κB to the degree observed with therapies targeting upstream metabolic dysfunction. Accordingly, the pro-inflammatory vascular environment associated with insulin resistance remains largely unaltered.
Cardiovascular outcome data further reflect this limitation. While early observational and subgroup analyses suggested benefit, more contemporary evidence demonstrates modest and inconsistent effects on cardiovascular outcomes, particularly when compared to therapies that directly target inflammation or insulin resistance.
Additional considerations include the potential for nutrient depletion, particularly vitamin B12, with long-term use [32], as well as gastrointestinal intolerance in a subset of patients.
In the setting of euglycemic diabetes, where glucose levels are already within normal range, the use of metformin represents a treatment strategy directed at a downstream surrogate rather than the upstream disease process. It lowers what is visible, but does not correct what is driving the pathology.
Accordingly, metformin may have a role within a broader therapeutic framework, but it should not be viewed as a primary or sufficient intervention in patients whose dominant abnormality is insulin resistance with preserved glycemia.
In this context, the limitation is not that metformin fails to lower glucose—it is that glucose is not the problem.
Icosapent Ethyl: Modification of Plaque Biology
Icosapent ethyl is a highly purified form of eicosapentaenoic acid (EPA), an omega-3 fatty acid that provides compelling evidence that cardiovascular risk reduction can be achieved through direct modification of plaque biology rather than through lipid lowering alone.
In the EVAPORATE trial, all patients were maintained on stable statin therapy, yet those receiving placebo demonstrated progression of high-risk plaque features, including a 109% increase in low-attenuation plaque, a phenotype strongly associated with plaque vulnerability. In contrast, patients receiving icosapent ethyl experienced a 17% reduction in low-attenuation plaque, along with favorable changes in other non-calcified plaque components [14].
These findings are critical. They demonstrate that even in the presence of aggressive LDL lowering with statins, high-risk plaque biology may continue to progress—and that targeted intervention can reverse these changes independent of cholesterol reduction.
This concept is reinforced by REDUCE-IT, which demonstrated a 25% relative risk reduction in major cardiovascular events, despite minimal additional LDL lowering. The magnitude of benefit observed cannot be explained by lipid effects alone and instead supports a mechanism driven by anti-inflammatory activity, membrane stabilization, and modulation of plaque composition.
Importantly, post hoc analyses from REDUCE-IT further strengthen this argument. Among patients who had already achieved LDL levels <55 mg/dL on high-intensity statin therapy, the addition of icosapent ethyl was associated with an additional 34% relative risk reduction in cardiovascular events. This occurred despite the absence of meaningful LDL reduction, underscoring that cardiovascular risk persists beyond cholesterol and can be modified through pathways independent of lipid lowering.
Taken together, these data reinforce a central principle: cardiovascular risk is not defined by LDL alone, but by the underlying biology of inflammation, metabolism, and plaque phenotype.
If LDL were the disease, these outcomes would not be possible.
Colchicine: Downstream Inflammatory Suppression Without Metabolic or Lipid Modification
Colchicine further supports the role of inflammation as a causal driver of atherosclerosis. Through inhibition of the NLRP3 inflammasome and downstream cytokine signaling, it reduces cardiovascular events in both acute and chronic coronary disease, as demonstrated in COLCOT and LoDoCo2 [17,18].
However, colchicine operates downstream within the disease cascade. It suppresses inflammatory signaling but does not address the upstream metabolic drivers that initiate and sustain that inflammation, including insulin resistance and compensatory hyperinsulinemia.
Equally important, colchicine does not modify lipid metabolism or alter lipoprotein profiles. It has no meaningful effect on LDL cholesterol, triglycerides, or the structural lipid components that contribute to plaque formation and progression. As such, its benefit is not derived from altering the substrate of atherosclerosis, but from attenuating the inflammatory response to it.
This distinction is critical.
Colchicine reduces the expression of disease, but not its origin. The metabolic environment characterized by impaired insulin signaling, excess circulating insulin, and associated oxidative stress remains unchanged. Lipid deposition within the arterial wall may continue, and plaque burden may persist or progress, even as inflammatory signaling is partially suppressed.
Accordingly, colchicine represents an effective adjunctive therapy for reducing inflammatory risk, but it is not a comprehensive solution. It neither corrects the metabolic dysfunction that drives atherosclerosis nor modifies the lipid substrate upon which the disease is built.
Its role, therefore, is best understood within a layered therapeutic framework: targeting downstream inflammation while upstream metabolic and lipid abnormalities are addressed through complementary strategies.
Colchicine dampens the fire—but it does not remove the fuel.
Beyond LDL: Reframing the Role of Statins in a Cardiometabolic Model
Statins remain foundational in cardiovascular medicine and have consistently demonstrated reductions in cardiovascular events. However, their mechanism of action—and therefore their therapeutic reach—is primarily centered on lipid lowering. While this is important, it does not directly address the upstream drivers of cardiometabolic disease, namely insulin resistance, hyperinsulinemia, and chronic inflammatory signaling.
This distinction is critical.
Atherosclerosis is not a cholesterol storage disorder in isolation. It is a biologically active, inflammatory disease driven by metabolic dysfunction. Lowering LDL cholesterol may reduce one component of risk, but it does not correct the underlying environment in which the disease develops. Insulin resistance, endothelial dysfunction, oxidative stress, and inflammatory pathway activation—particularly through NF-κB signaling—continue to operate despite aggressive lipid lowering [5,7,21–23].
Imaging studies have consistently demonstrated this disconnect. While statins may modestly reduce plaque volume or stabilize certain plaque features, they do not uniformly reverse high-risk plaque biology. Residual inflammatory and metabolic risk persists, and disease progression can continue even in the setting of well-controlled LDL levels [4,14,15].
The EVAPORATE trial provides particularly important insight into this limitation[14]. In this study, all patients were on stable statin therapy, yet those receiving placebo in addition to statins demonstrated progression of high-risk plaque features, including a 32% increase in fibrofatty plaque and a 109% increase in low-attenuation plaque over approximately 18 months . These are the very plaque phenotypes associated with vulnerability and acute coronary events.
In contrast, the addition of icosapent ethyl led to a 34% reduction in fibrofatty plaque and a 17% reduction in low-attenuation plaque, despite minimal additional LDL lowering. This divergence highlights a fundamental concept: plaque biology is not determined by LDL alone.
The persistence—and in some cases progression—of high-risk plaque despite statin therapy underscores the limitation of a lipid-centric model. Statins do not directly improve insulin sensitivity, do not meaningfully reduce hyperinsulinemia, and exert only modest and indirect effects on inflammatory signaling. As a result, the upstream metabolic environment that drives atherosclerosis remains largely unaltered.
This helps explain a commonly observed clinical paradox: patients with “optimal” LDL levels who continue to experience cardiovascular events. The disease is not being missed—it is being incompletely addressed.
Accordingly, statins should not be viewed as comprehensive therapy for atherosclerosis, but rather as one component of a broader, biology-driven strategy. When used in isolation, they target a downstream manifestation of disease. When integrated into a framework that also addresses insulin resistance, inflammation, and metabolic dysfunction, they become part of a more complete and effective approach.
The shift, therefore, is not away from statins, but away from statin-centric thinking.
Lowering LDL may reduce risk—but it does not redefine the disease. Understanding the biology does.
These therapies do not compete—they map onto different levels of the disease process. Atherosclerosis is not driven by a single pathway, and no single intervention fully addresses its complexity. Insulin resistance and hyperinsulinemia define the upstream metabolic environment. Lipid accumulation and plaque composition represent the structural substrate. Inflammatory signaling governs plaque activity and instability. Accordingly, therapies that target these domains operate at different, but complementary, levels of disease biology. Pioglitazone addresses the metabolic origin, icosapent ethyl modifies plaque phenotype and inflammatory tone, colchicine suppresses downstream inflammatory expression, and statins reduce lipid burden. When viewed through this framework, the goal is not to choose between therapies, but to align them with the mechanisms driving disease in each individual patient.
Normal Glycemia, Pathologic Physiology: A New Framework for Understanding and Action
The metabolic data presented here reveal a fundamental truth: normal glucose does not indicate normal physiology. Insulin resistance is the upstream driver, hyperinsulinemia sustains metabolic stress, inflammation propagates vascular injury, and plaque represents the structural endpoint. Insulin reveals the burden. Imaging reveals the consequence.
But recognizing this model is only the beginning. The critical next step is understanding why this biology develops in the first place.
At CardioCore Metabolic Wellness Center, this is where the approach fundamentally shifts. We move beyond surface-level diagnostics and into a functional cardiometabolic evaluation designed to uncover the drivers of disease. This includes assessment of genetic predispositions that influence metabolism, inflammation, and vascular risk. It includes advanced metabolic profiling to understand which fuels the body is using, how efficiently energy is being produced, and whether detoxification and mitochondrial pathways are functioning optimally. Hormonal evaluation is central, as cortisol dysregulation, testosterone deficiency, and broader endocrine imbalances directly influence insulin resistance and inflammatory signaling.
Equally important is the gut microbiome, which plays a critical role in metabolic regulation, immune activation, and systemic inflammation. Disruption at this level can amplify insulin resistance and vascular disease in ways that are often invisible to conventional testing.
This biologic understanding is then integrated into a comprehensive, personalized strategy that includes nutrition as information, targeted supplementation, structured exercise, identification of carbohydrate intolerance, stress regulation, sleep optimization, and the often-overlooked domains of socialization and behavioral health. These are not adjuncts to care. They are the foundation of it.
“Because the most important question is not whether a patient needs a stent, it is why the disease developed in the first place.”
The most important question is: Why did the disease develop in the first place, and what variable are we going to change to prevent it from happening again?
Even in patients who have undergone stenting or bypass surgery, the underlying biology remains. If that biology is not identified and addressed, the disease does not end. It progresses.
The old model asks whether there is blockage.
The new model asks what is driving the disease.
Fool me once, shame on you. Fool me twice, shame on me. Or as Einstein described it, doing the same thing over and over again and expecting a different result is the definition of insanity.
At CardioCore, we choose a different path. We think differently. We look where others are not looking. And we identify the drivers of cardiovascular disease—even when they are hidden in plain sight.
“If we continue to treat glucose, we will continue to miss the disease. If we begin to treat the biology, we may finally change its course.”
References (Final)
Jesús M, et al. Am J Cardiol. 2013;111:333–338.
CTT Collaboration. Lancet. 2010;376:1670–1681.
Sattar N, et al. Lancet. 2010;375:735–742.
Nissen SE, et al. JAMA. 2006;295:1556–1565.
Steinberg HO, et al. J Clin Invest. 1996;97:2601–2610.
DeFronzo RA. Diabetes. 2009;58:773–795.
Reaven GM. Diabetes. 1988;37:1595–1607.
Marx N, et al. Circ Res. 2004;94:116–123.
DeFronzo RA, et al. Diab Vasc Dis Res. 2019;16:133–143.
Budoff MJ, et al. Eur Heart J. 2020;41:3925–3932.
Ridker PM, et al. N Engl J Med. 2017;377:1119–1131.
SCOT-HEART. N Engl J Med. 2015;372:2343–2352.
Cury RC, et al. J Cardiovasc Comput Tomogr. 2022;16:536–557.
Tardif JC, et al. N Engl J Med. 2019;381:2497–2505.
Nidorf SM, et al. N Engl J Med. 2020;383:1838–1847.
Hall KD, et al. Lancet. 2012;379:130–140.
Hotamisligil GS. Nature. 2006;444:860–867.
Tilg H, et al. Nat Rev Immunol. 2008;8:772–783.
Shoelson SE, et al. J Clin Invest. 2007;116:1793–1801.
Ebbeling CB, et al. N Engl J Med. 2012;367:2489–2499.
DeFronzo RA, Tripathy D. Diabetes Care. 2009;32:S157–S163.
Holloszy JO. J Biol Chem. 2008.
Abdul-Ghani MA, et al. Diabetes Care. 2006;29:1130–1139.
24.Dimitriadis G, Mitrou P, Lambadiari V, et al. Insulin effects in muscle and adipose tissue. Endocr Rev. 2011;32(4):533–582.
25.Service FJ. Hypoglycemic disorders. N Engl J Med. 1995;332(17):1144–1152.
26.Ferrannini G, De Bacquer D, De Backer G, et al. Dysglycaemia screening and its prognostic impact in patients with coronary artery disease: EUROASPIRE IV and V. Lancet Diabetes Endocrinol. 2024;12(1):XX–XX.
27.Randle PJ, Garland PB, Hales CN, Newsholme EA. The glucose fatty-acid cycle: its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet. 1963;1(7285):785–789.
28.Yki-Järvinen H. Thiazolidinediones. N Engl J Med. 2004;351(11):1106–1118.
29.Miyazaki Y, Mahankali A, Matsuda M, et al. Effect of pioglitazone on abdominal fat distribution and insulin sensitivity in type 2 diabetes mellitus. J Clin Endocrinol Metab. 2002;87(6):2784–2791.
30.Miller RA, Birnbaum MJ. An energetic tale of AMPK-independent effects of metformin. Cell Metab. 2019;29(3):507–508.
31.Konopka AR, Laurin JL, Schoenberg HM, et al. Metformin inhibits mitochondrial adaptations to aerobic exercise training in older adults. Aging Cell. 2019;18(1):e12880.
32.de Jager J, Kooy A, Lehert P, et al. Long term treatment with metformin in patients with type 2 diabetes and risk of vitamin B12 deficiency: randomised placebo controlled trial. BMJ. 2010;340:c2181.
